
FDA Warns Against Using Homeopathic Cures Especially On Babies

A full-time house-wife based in Colorado Springs, Karina Talbott, gave birth to Blaine Talbott as a healthy baby and at two months old, he began teething. As a caring mother, she went in search of a reliever that would be mild and safe for her son. She found a teething drug called homeopathic at the pharmacy and decided to try it on, Blaine.
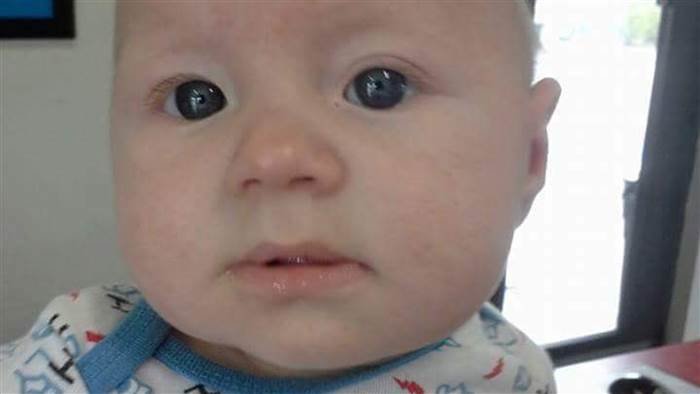
Blaine started convulsing after his mom administered the drug to him.
She was attracted by its description which says that the drug was completely natural so she thought it would be the best choice for Blaine.
Unfortunately, not sooner than she administered the drug to Blaine that he started convulsing. However, she was advised to pause the homeopathic medication, and when she did, the convulsion ceased.
Blaine survived the episode and now has clocked four years old. Despite the end to his convulsion, Karina reported that he seems to be undergoing certain setbacks in mental development, but she couldn’t decipher whether the homeopathic tablets caused this. When she discovered this, she sent complaints to the manufacturers Hyland. They responded by asking for the product number, and she sent it to them. Till date, Karina is yet to get their feedback.
This is the case with many American families. The Food and Drug Administration has started a fierce campaign against the use of homeopathic teething tablets because, according to findings, it has belladonna, a poisonous species of plant.
The FDA is ready to bring down the homeopathy cartel that is estimated to worth around $3 billion annually. FDA has the power to control the production of homeopathic drugs, but it has lagged behind in that regard for some time.
In a statement issued by the agency, they have decided to adopt a risk-based reinforcement system to curtail the spread of homeopathic tablets.
This is geared towards shielding consumers from getting harmed by the tablets, even if they chose to patronize the products. This planned system would renew the FDA’s already extant policy aimed at improving the handling of situations where homeopathic medications are being sold to treat severe diseases and sicknesses. Investigations will be made on whether the drugs have positive health effects or not.
Going further, the agency explained that the reinforcement would also reach out to circumstances that may depict some unhealthy constituents mixed in making homeopathic products. 
Adding to the voice of FDA, one of the subsidiaries of the National Institutes of Health, the National Center for Complementary and Integrative Health, also argued that there is lack of scientific evidence for homeopathy to show that it is a good drug worthy of recommendation for diseases and illnesses. This is due to many major theories used in homeopathy that are in line with fundamental principles of physics and chemistry.
The worst discovery in homeopathic products is the use of expired ingredients. This is because homeopathy itself was manufactured as back as the 18th century where there was an inadequate medical, scientific advancement.
Confirming this historical fact, a medical expert in infectious diseases at Children’s Hospital in Philadelphia and an author, Dr. Paul Offit, noted that there were very few drugs for several ailments in those days. Examples are willow bark made from aspirin and quinine to tackle malaria.
The secret behind homeopathy was that it was originally produced as an optional drug to be administered to patients who are stooling and bleeding. The concept is based on soaking something inside water and allowing the H2O to identify with the substance. This unreliable method is being adopted to produce dangerous drugs in a seemingly safe way to deceive consumers.
However, recorded list of surgery-related processes, medications, and vaccines have passed the safety standard, certifying them safe for use but over two centuries later, homeopathy is yet to make that list.
Offit declared that when homeopathic is soaked in water, the consumer ended up using the dangerous substance.
Commenting on the planned risk-based reinforcement, the Commissioner for FDA, Dr. Scott Gottlieb, said that the proposal is obligatory and the general public is free to also give their thoughts on it.
He noted that the agency is not seeking ways to stop some people from choosing to use optional medications. However, FDA must perform its duty in ensuring that people are free from drugs that may be useless to them or even worse, harm them.
In January 2016, FDA ordered Hyland to release official instruction to its consumers to discard the empty bottles of used homeopathic drugs that contained hints of belladonna. This didn’t go down well with Hyland, but they succumbed much later. In October, the company ceased the production and marketing of the tablets.
FDA is also on the verge of abolishing a deal signed in 1988 which exempted homeopaths from the official FDA approval standard with the hope that they would obey manufacturing rules and regulations.
Despite all these plans, the Commission has decided to take it one step at a time. Only suspected dangerous homeopaths will be faced at first.
More in Medical Conditions
-
A Comprehensive Guide on How to Heal Anxious Attachment Style
If you often find yourself seeking constant reassurance from your partner or requiring endless validation to feel secure, you may be...
June 1, 2024 -
Why Is My Skin Peeling on My Face After Skincare? Here’s What You Need to Know
Experiencing skin peeling on your face after implementing a skincare routine can be perplexing and frustrating. This phenomenon, where the skin...
May 23, 2024 -
What Is a Mental Edge, and How Do You Get It?
Have you ever wondered why some individuals seem unshakably confident under intense pressure, finding a way to excel when it matters...
May 15, 2024 -
When to Worry About Varicose Veins? Here’s What You Need to Know
Varicose veins are a common issue, affecting about 20% of adults. They’re usually seen as unsightly blemishes on the legs but...
May 12, 2024 -
Why Is Discipline Important: The Ultimate Guide to the Importance of Discipline
Discipline is undoubtedly one of life’s most crucial character traits. It shapes how we approach tasks, manage our time, and...
May 3, 2024 -
Have I Fallen Out of Love or Am I Depressed? Everything You Need to Know
Have I fallen out of love or am I depressed? You’re not alone in this query. In the diversity of human...
April 26, 2024 -
What Does Lung Cancer Breath Smell Like?
Cancer is one of the most formidable diseases of our time, characterized by the uncontrolled growth of cells that invade and...
April 20, 2024 -
Making Major Life Changes? Discover 5 Crucial Things Before You Leap
Life changes are a pivotal part of our journey, marking the transition from one phase of life to another. These changes,...
April 13, 2024 -
The Surprising Benefits of Being Bored at Work
Are you bored at work? We’ve all been there, staring blankly at our computer screens, feeling an overwhelming urge to be...
April 6, 2024














You must be logged in to post a comment Login